Children’s Hospital Los Angeles Offers MIBG Therapy for Neuroblastoma
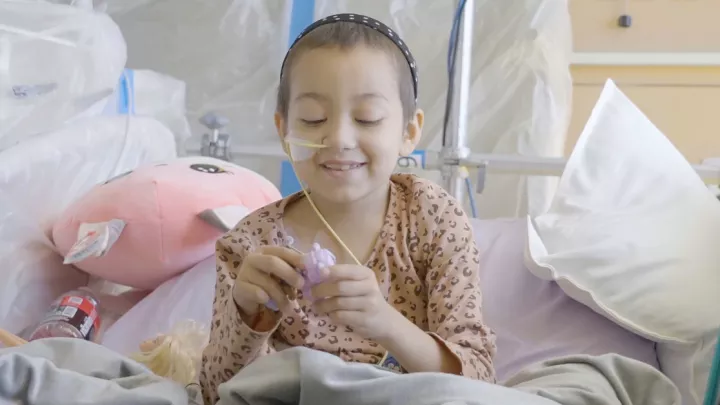
Children with high-risk neuroblastoma can now access a specialized, targeted radiation treatment called MIBG therapy at Children’s Hospital Los Angeles—one of the largest neuroblastoma programs in the country and the only pediatric facility in Southern California and the Southwestern United States to offer this treatment.
MIBG (metaiodobenzylguanidine) is a compound that was once a blood pressure medicine and is easily absorbed by neuroblastoma cells. In MIBG therapy, this chemical is combined with a radioactive iodine called I-131 and given to patients through an IV infusion, allowing it to kill tumor cells throughout the body.
Neuroblastoma is the second-most common solid tumor in children (after brain tumors). The cancer develops from immature nerve cells and typically affects children between the ages of 2 and 4. Nearly half of patients are diagnosed with high-risk, metastatic disease, which has a 50% mortality rate.
“Bringing MIBG therapy to Children’s Hospital Los Angeles will allow many more patients to access this promising treatment,” says Araz Marachelian, MD, MS, Medical Director of the Neuroblastoma MIBG Program in the Cancer and Blood Disease Institute at Children’s Hospital Los Angeles. “Previously, families in Southern California, as well as nearby states like Arizona and Nevada, have had to travel long distances for MIBG treatment. We are excited to give them an option much closer to home.”
Alan S. Wayne, MD, Director of the Cancer and Blood Disease Institute, adds that the Neuroblastoma team at Children’s Hospital Los Angeles is dedicated to developing the most advanced and promising new therapies for children with high-risk neuroblastoma. “MIBG therapy is one more example of our commitment to provide the highest quality cancer care to children from Southern California and around the globe,” he says.
Parents can stay with their child
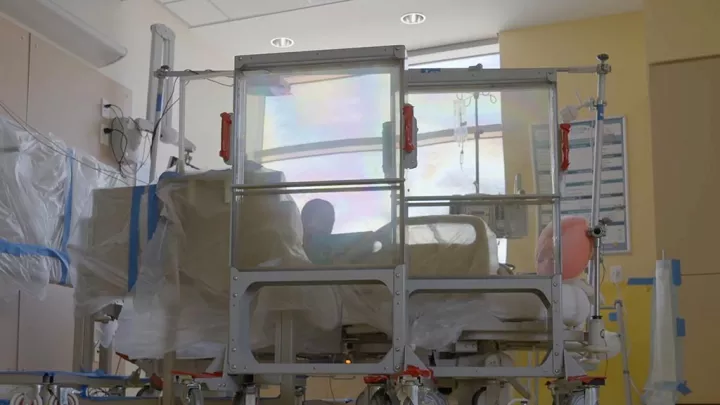
Patients receive MIBG therapy while in the hospital, staying in a special room specifically designed for this treatment. After a one-time infusion, which takes about two hours, children must remain in the room for two to five days, or until radiation levels in their body are low enough for them to safely go home.
Children’s Hospital Los Angeles—which began offering MIBG therapy in March—is one of only a few institutions in the country to enable parents to stay in the same room with their child during the treatment. This is possible because the room is very large, allowing parents to be a safe distance from radioactivity.
The room also contains extensive radiation shielding, including special shields on wheels around the patient’s bed. These use an innovative clear material—an extremely dense liquid encased in plexiglass—that allows the parent and child to see each other at all times. Parents can also be at their child’s bedside for short periods, with monitors tracking their radiation exposure.
“Distance is very effective in stopping radiation,” says Chuck Pickering, Vice President of Facilities and Support Services at Children’s Hospital Los Angeles. “By using a combination of distance, shielding and monitors, we are able to keep parent radiation exposures to very low, safe levels, enabling them to be with their child.”
Multiple clinical trials
MIBG therapy is not approved by the Food and Drug Administration and is being studied in multiple clinical trials at Children’s Hospital Los Angeles and select sites around the country.
One of these trials, led by the Children’s Oncology Group national consortium, is a phase 3 randomized controlled trial studying the effectiveness of MIBG when added to standard therapy for newly diagnosed patients with high-risk neuroblastoma.
And while MIBG therapy has long been given to children with relapsed or treatment-resistant disease, another multicenter trial is evaluating a novel combination for those patients: giving MIBG along with immunotherapy. That phase 1 trial is led by the New Approaches to Neuroblastoma Therapy (NANT) consortium, which is headquartered at Children’s Hospital Los Angeles.
For more information on MIBG therapy, read a post on chla.org and call 323-361-4100.
About Children’s Hospital Los Angeles
Founded in 1901,Children’s Hospital Los Angeles is the largest provider of care for children in Los Angeles County. Families travel from all 50 states and more than 90 countries so that their children can receive the very best care. The hospital’s clinical teams treat patients ranging in age from newborn infants to young adults for everything from well-child visits to organ transplantation. It is ranked the top children’s hospital in California as well as in the Pacific region, and fifth in the nation for clinical excellence by U.S. News & World Report in its prestigious Honor Roll of Best Children’s Hospitals. Clinical care is led by physicians who are faculty members of the Keck School of Medicine of USC through an affiliation dating from 1932. It is home to the largest pediatric residency training program at a freestanding children’s hospital of its kind in the western United States. Among top 10 children’s hospitals for National Institutes of Health funding, The Saban Research Institute of Children’s Hospital Los Angeles leads the field in innovative bench-to-bedside basic, translational and clinical research conducted in pediatrics. To learn more, follow us on Facebook, Instagram, LinkedIn, YouTube and Twitter, and visit our blog at CHLA.org/blog.