Guy Young, MD
Following medical school, I completed my residency in pediatrics at Schneider Children's Hospital-Long Island Jewish Medical Center which is part of the Albert Einstein College of Medicine. I then completed a fellowship in pediatric hematology/oncology at Children's National Medical Center in Washington, DC which is part of the George Washington University School of Medicine.
I then began my research career at the Children's Hospital of Orange County under the mentorship of Dr. Diane Nugent. There, I became interested in improving the treatment for patients with hemophilia and inhibitors and for children with blood clots.
I received my first independent grants there inculding a grant from the FDA to study a new anticoagulant in children. In addition, I became interested in thromboelastography.
In 2007, I moved the Children's Hospital Los Angeles where I am continuing to perform research in the same areas as I had started in Orange County.
Pediatric Thrombosis Hemophilia
Education
SUNY at Stony Brook
Schneider Children's Hospital at Long Island Jewish Medical Center
Schneider Children's Hospital at Long Island Jewish Medical Center
Children's National Medical Center
Accomplishments
Pediatrics; Pediatric Hematology/Oncology
American Society of Hematology/Oncology American Society of Hematology World Federation of Hemophilia International Society on Thrombosis and Haemostasis Hemophilia and Thrombosis Research Society
American Society of Cancer Clinical Fellowship Award
Publications
Research
Our research focuses on state-of-the-art therapies for children with bleeding or clotting disorders.
- We provide children the opportunity to receive the latest available treatments for coagulation disorders.
- We also provide families the option of participating in clinical trials.
The Coagulation Research Lab conducts three types of research:
- Clinical trials evaluating new drugs for children with hemophilia.
- Clinical trials evaluating new drugs for children with thrombosis.
- Laboratory research utilizing thromboelastography, a device that measures clot formation over time. The goal of this research is to develop new methods for predicting which patients are at highest risk for developing thrombosis and for determining the effects of drugs that help promote or prevent clot formation.
The main thrust of our research is to improve the management of patients with hemophilia and thrombosis. This involves testing novel therapies and assessing the response to those therapies.
In hemophilia, patients who develop antibodies to treatment cannot be treated with conventional clotting factor replacements and they suffer the worst consequences of the disease. Through collaboration with industry, we are testing several new compounds to improve patient outcomes.
In patients with thrombosis, a condition that is increasingly common in children, we are testing several drugs that have been approved for adult use but still need to be demonstrated as safe and effective in pediatric patients. These drugs have improved properties over standard anticoagulants.
Visit the Young Laboratory.
Thromboelastography curve showing the effect of a medication (FEIBA) on the blood of a child with hemophilia.
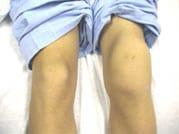
A child experiencing bleeding in his right knee. This is the most common symptom of hemophilia.
One of the major gaps in coagulation research is the absence of a clinically useful test to predict bleeding or clotting events and to predict the outcome of interventions (medications). Our lab has been using a device called the thromboelastograph for this purpose. The thromboelastograph is allowing us to develop a test to predict thrombosis. While several categories of patients are known to be at high risk for thrombosis, predicting which of those patients will actually get a clot is not yet possible.
By using a relatively simple blood test, our goal is to predict, with a high degree of accuracy, which patients will form a blood clot. This will in turn allow us to select patients in whom to intervene with medications in order to prevent thrombosis. The testing and prophylactic medication is similar to the process used in adults for cholesterol.
GOALS
Our goals are to continue bringing state-of-the-art treatments and technologies to our research program and to offer patients the most novel approaches available. Our team has been successful in obtaining a variety of funding sources to support our research. These include federal grants from the National Institutes of Health (NIH), the Centers for Disease Control and Prevention (CDC), and the Health Resources and Services Administration (HRSA), as well as private foundation grants and pharmaceutical grants and contracts. Several influential publications have resulted from this work and have influenced clinical practice.
KEY FINDINGS
Our study of new anticoagulants (fondaparinux, bivalirudin and argatroban) and our demonstration of their safety and effectiveness compared to standard treatments, has benefited children throughout the world.
We have changed the approach for the management of patients with hemophilia by introducing novel regimens for the use of existing drugs while awaiting the completion of studies using new agents.
We have also demonstrated the utility of doing research with the thromboelastograph. Prior to our publications over the past several years, only three centers in the United States had access to this device. Now, over 20 research centers have acquired this technology for use in research. We anticipate that this device will soon begin to be used in the clinical setting.
CURRENT FUNDING
Our current funding includes NIH, CDC and HRSA grants. In addition, we have a grant from a philanthropic organization, the Oxnard Foundation, as well as pharmaceutical company funding.
NIH: 1R01HL095110
Guy Young - Principal investigator (PI)
This is a multi-center, open-label clinical trial aimed at determining the dosing, pharmacokinetics, safety and efficacy of bivalirudin, a direct thrombin inhibitor in children with venous thromboembolism.
Role: PI
DHHS-HRS: 6H30MC0036
Diane Nugent - PI
Comprehensive Hemophilia Care
The study uses multidisciplinary teams to provide comprehensive diagnosis, assessment, surveillance, outreach, education, consultation and prevention services to people with bleeding disorders that are directed at attaining and measuring specific outcomes to reduce complications.
Role: Co-investigator
Oxnard Foundation
Guy Young - PI
Developing Thromboelastography as a Clinical Tool for Detection of Thrombosis.
Role: PI
American Thrombosis and Hemostasis Network
Guy Young - PI
Data Management Support for ATHNdataset
Media
Medscape Medical News - Hemophilia A: Bleeds Plummet After Experimental Gene Therapy
HCP Live - Guy Young, MD: Prophylactic or Gene Therapy, and Shared Decision Making in Hemophilia
Managed Healthcare Executive - Fitusiran Emerges as a Potential Pan-Hemophilia Rebalancing Agent
MD Magazine — Extending Time Between Infusions for Pediatric Hemophilia
Yahoo Health- New Hemophilia Drugs Aim to Simplify Treatment
Science Daily — Disruptive technology for the treatment of hemophilia
US News & World Report - Another Gene Therapy Breakthrough Against Hemophilia
CHLA Blog
CHLA Experts to Present at 2024 American Society of Hematology Meeting