Heart Institute Research
Director: Vaughn A. Starnes, MD
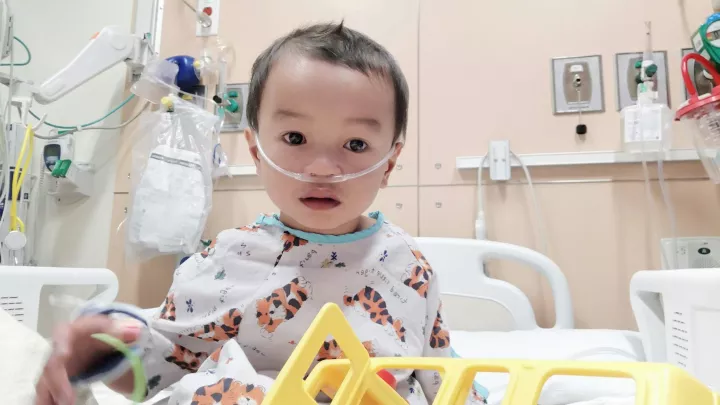
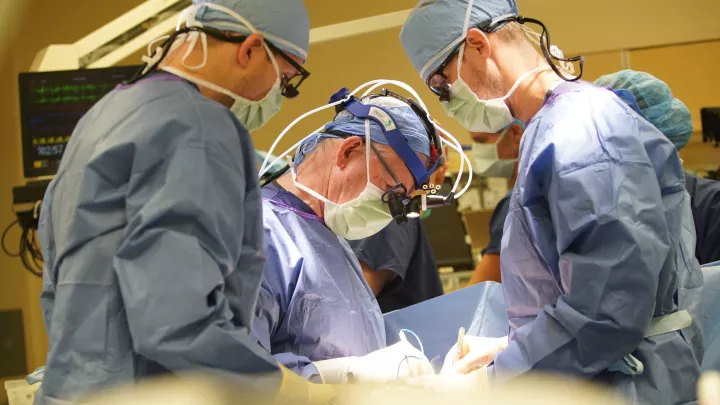
In the first-ever randomized surgical trial for pediatric heart disease, cardiac surgeons and cardiologists are asking how to ensure the optimal survival of babies born with hypoplastic left heart syndrome (HLHS). “This kind of study takes a tremendous commitment. It’s the only way to truly answer such a complex question,” says Alan B. Lewis, MD, co-principal investigator at Children's Hospital Los Angeles with Vaughn A. Starnes, MD.
The Single Ventricle Reconstruction (SVR) Study, funded by the National Heart, Lung and Blood Institute of the National Institutes of Health, involves Children's Hospital Los Angeles and 14 other members of the Pediatric Heart Network, a consortium of major pediatric cardiac institutions.
In HLHS, which is fatal without corrective surgery, the left ventricle is missing or underdeveloped. The study’s goal: determine which of two shunt positions, if either, offers advantages for patients undergoing the Norwood Procedure, the first stage in repairing a single ventricle heart. It involves enlarging the aorta, opening the wall between upper chambers of the heart and inserting a shunt or small connector to deliver blood to the lungs.
Each child in the study is randomly assigned a shunt position. The surgeon can, however, override that decision. Participants will be followed for 14 months, with plans for long-term follow-up, given funding. Research Coordinator Nancy Pike, RN, PNP, PhD, is working to enroll 35 families at Children's Hospital Los Angeles by this fall (out of a total of 466 nationwide).
Research Focus
Clinical Research is performed within The Heart Institute and directed primarily by Drs. Vaughn Starnes, the Chair of Cardiothoracic Surgery at Children's Hospital Los Angeles and at USC, and by his department members Drs. Mark Barr, Winfield Wells and John Nigro. Dr. Starnes is involved in a number of Clinical trials, such as barrier films to prevent adhesions during open-heart surgery (with Dr. Wells) and the use of Viagra to treat pulmonary hypertension (with colleagues in Cardiology). Dr. Nigro is exploring new surgical techniques for the repair of congenital cardiopulmonary defects in neonates, and Dr. Barr is working with Dr. Wu to advance organ preservation technologies and to develop new strategies for the treatment and prevention of chronic rejection in heart and lung transplant patients.
Cardiac Research
In spite of the advances made in pediatric cardiac disease, much remains to be done. About 32,000 infants are born each year in the United States with congenital cardiovascular defects; others acquire such conditions after birth.
Physician-scientists in the Heart Institute at Children's Hospital Los Angeles are engaged in research to improve the odds of transplantation success, advance the capabilities of imaging, refine treatments for Kawasaki Syndrome, and develop new medications for primary pulmonary hypertension, among other topics. The Institute works to forge a strong link between clinical care and basic research, so that the best ideas can reach patients’ bedsides as quickly as possible.
About our Laboratory Research Efforts
Laboratory Facilities
The Heart Valve Laboratory is located on the fourth floor of the The Saban Research Institute at Children's Hospital Los Angeles. It consists of adjoining wet lab space, equipment rooms, tissue culture facilities and laboratories for computer modeling and materials testing.
The laboratory is contiguous with that of other scientists working on organ transplantation and fibrosis, thus enhancing collaborative activities.
The materials testing facility is contained in an adjoining equipment room and is equipped with an Instron 5843 single column material testing system, hydrodynamic valve tester, flexural fatigue tester and a small drill press/milling machine for small parts fabrication. The Instron is a horizontal unit, designed for high precision/low force testing of small tissue specimens, such as our tissue-engineered materials.
Microscopy facilities consist of 3 Olympus research microscope systems with digital camera Microfire. The three microscopes are ZX9 stereomicroscope system, BX51 upright microscope system with the capability of polarized light and fluorescence, and CKX41 inverted live cell microscope system.
Through collaborative projects, we make use of the Histopathology and Electron Microscopy Laboratory core facility in the Department.
Resources
We work with the following heart valve companies for our research endeavors:
Additional Resources
- Medical Device Link -- Provides news and information about the medical device industry
- Composites World -- Provides a Sourcebook directory of products and services for the composites industry
- Association for the Advancement of Medical Instrumentation
- ASTM International
- American National Standards Institute
- Food and Drug Administration
- Society for Biomaterials